In this final lesson of the unit students are tasked with reviewing for an upcoming acid-base test. Their activities include working on a practice test, getting help from the teacher, and each other, and playing acid-base Jeopardy.
This lesson aligns to the NGSS Practices of the Scientist of Using mathematics and computational thinking because much of the study guide has a mathematical component to it. It also aligns to the practice of Obtaining, evaluating, and communicating information because at this point in the unit students are processing and synthesizing the information they have been working with throughout the unit. It aligns to the NGSS Crosscutting Concept of Structure and Function because students are coming to an understanding of the properties of acids and bases by studying their molecular substructures. Some of this lesson also aligns to the HS-PS1-7: Use mathematical representations to support the claim that atoms, and therefore mass, are conserved during a chemical reaction because students must use a balanced chemical equation in order to calculate the molarity correctly and show how neutralization reactions work. In terms of prior knowledge or skills, students should be well along in their understanding of acids and bases by this point. There are no special materials needed for this lesson, but having access to multiple computers for the Jeopardy game was quite useful in my classroom. Alternatively, the whole class could play, but this creates its own challenges in terms of to what extent each student is participating.
Do Now: To start class I ask students to do one of two things, depending on how ready for class they are. If they have completed the, I ask them to find a partner and compare answers. If they have not completed the practice test, I ask them to continue working on it. I reason this is a good way to start class because it will launch students into the learning activity for today, which is to prepare for the upcoming acid-base test. During this time I walk around and observe how well students have prepared for the exam. I learn that some students need to still buckle down and work on the practice test, while others are ready for other learning activities that will help them prepare for the test. Mini-lesson: The mini-lesson today is quite short because this is a student-directed class.
I do not teach content today; rather, I make sure that I articulate clear expectations for what is happening in class today. I start by reminding students that their test is coming up and today is designed for students to work on clarifying things that have confused them throughout the unit.
I explain that students should first finish the practice test. When they have finished they need to show me the completed practice test and I will give them the answer key. They will check their work and then tell me how they did. If they did well, I ask them to sign out a laptop and download the Jeopardy game from my website. If they do not do well I ask them to review their work with the goal of understanding why they got certain questions wrong. I also note that I am available for conferencing throughout the class.
To check for understanding I then ask a student to summarize for the class what the chain of events will be in today's class. Student Activity: Once I am satisfied that students understand my expectations for today’s class, I invite them to get to work.
Several students approach me for the. I briefly check their work for completeness and distribute the answer key to them, checking their names off on checklist so I can keep track of who is making progress on the day’s agenda. Other students are busy working on the practice test. Once everyone has an initial task, I check in with the whole class to let them know that I am available for individual questions. This is a good example of what this looks like.
As members of the initial group of students check in with me about their work, I check in with them about how they did and listen to their explanations for questions they got incorrect. I then authorize them to sign out a laptop once they have shown proficiency with the practice test so that they can play.
Students play this game in groups of 2-3. I keep a small stash of candy on hand so that students can compete for it during the game, making it just a bit more lively.
The Jeopardy game is a fun way to review material. The template I use is downloaded from. Some tips on making the game work: to get back to the home screen, click the bottom right-hand corner of the slide. Do not save the program while you are playing it, and that way the squares on the home screen will be the correct color based on whether the question has been asked. I have structured my class this way because I do not think review sessions should be passive events.
I think students should be actively working to advance their thinking, and they can do that best by engaging in activities that force them to think. Catch and Release Opportunities: About half way through the class I report that about half the students have completed the practice test, and that several more students are almost ready for the answer key, such as the owner of this. I do this to show that there is momentum in the room, and this can be encouraging for students. Most students want to feel like they have accomplished something, and reminding them of the flow of events helps them fulfill this desire. To wrap this lesson up I report out our final numbers.
Ninety percent of students have completed the study guide. I note that this is a good start. However, I also note that completing the study guide is not the goal. The goal is to be able to take a test similar to the study guide with no notes or help from others.
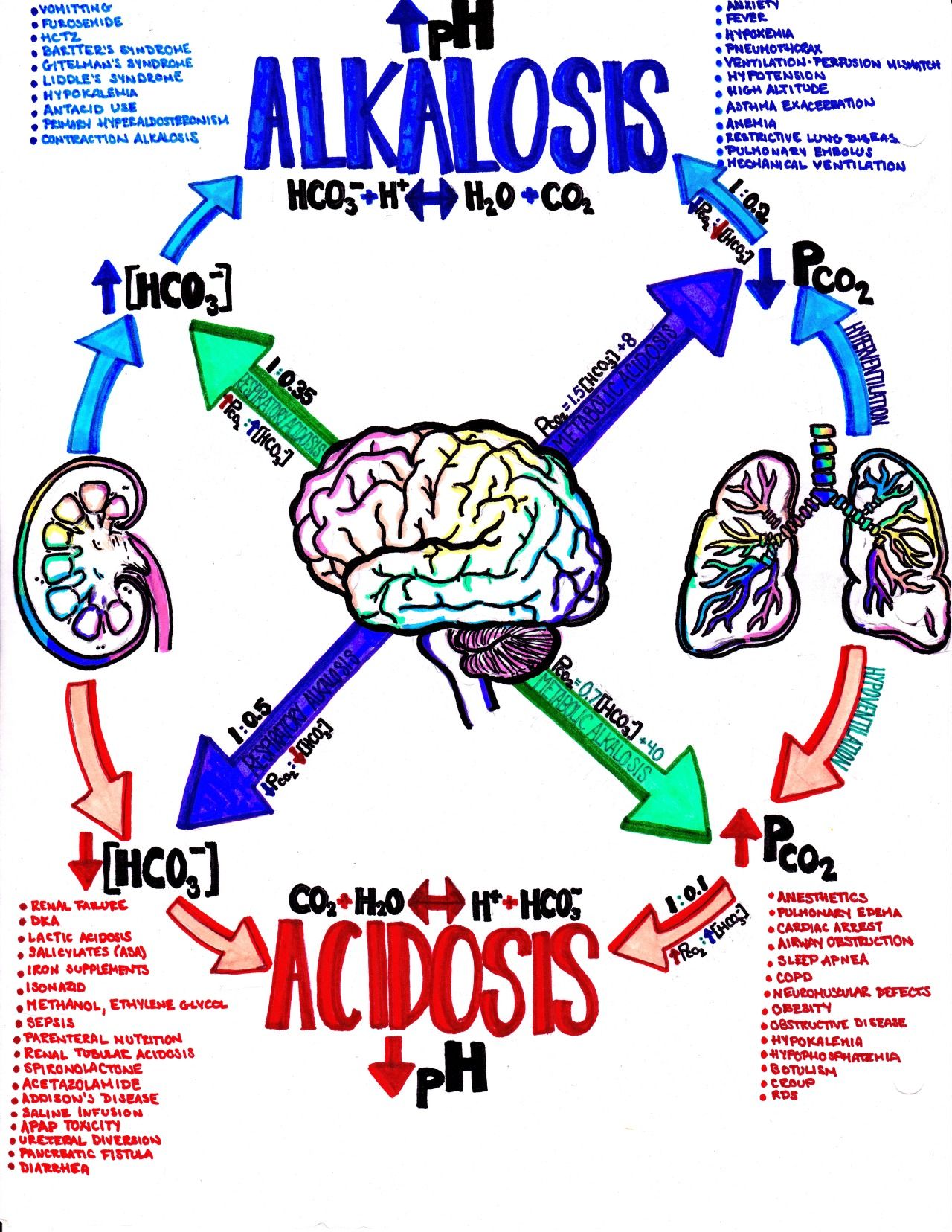
Toward this latter goal, I ask that students look over the practice test for homework and identify things that they are struggling with. I ask them to identify things they need to remember for the test, and use flash cards or some other device to commit those things to memory.
Acid Base Equilibria MCQs Quiz Online Practice, A level chemistry test for online courses learning and test prep. Has multiple choice questions (MCQ), acid base equilibria quiz questions and answers to learn. GCE chemistry practice test MCQ on on description of proton, new classification of acid and base was given by with options j.bronsted, t.lowry, j.dalton and both a and b problem solving skills for viva, competitive exam prep, interview questions with answer key. Free study guide is for online learning with MCQs to practice test questions with answers.
Review: Acid and Base Strength Why is it that some acids can eat through glass, but we can safely consume others? Why can you cook with a base like baking soda, but you should be extremely cautious when handling a base like drain cleaner? The answer lies in the ability of each acid or base to break apart, or dissociate: strong acids and bases dissociate well (approximately 100% dissociation occurs); weak acids and bases don't dissociate well (dissociation is much, much less than 100%). Let's go to the lab and zoom into a sample of hydrochloric acid to see what's happening on the molecular level. Given that hydrochloric acid is a strong acid, can you guess what it's going to look like inside?
Hydrochloric Acid There are no HCl molecules to be found because 100% of the HCl molecules have broken apart into hydrogen ions and chloride ions. In fact, the hydrogen ions have attached themselves to water to form hydronium ions (H3O+). We would write out the dissociation of hydrochloric acid as HCl + H2O - H3O+ + Cl. HCl is the parent acid, H3O+ is the conjugate acid, and Cl- is the conjugate base.

The conjugate acid and conjugate base occur in a 1:1 ratio. In case it's not fresh in your mind, a conjugate acid is the protonated product in an acid-base reaction or dissociation. A conjugate base is the negatively charged particle that remains after a proton has dissociated from an acid. If we were to zoom into our sample of hydrofluoric acid, a weak acid, we would find that very few of our HF molecules have dissociated.
Lord Of The Flies Study Guide Answers
However, we would still write the dissociation the same: HF + H2O - H3O+ + F. In fact, for all acids we can use a general expression for dissociation using the generic acid HA: HA + H2O - H3O+ + A. For all bases, we can use a general equation using the generic base B: B + H2O - BH+ + OH. B is the parent base, BH+ is the conjugate acid, and OH- is the conjugate base. We know what is going on chemically, but what if we can't zoom into the molecular level to see dissociation?
How is acid or base dissociation measured then? Intro to Ka and Kb We use dissociation constants to measure how well an acid or base dissociates. For acids, these values are represented by Ka; for bases, Kb.
These constants have no units. All chemical reactions proceed until they reach chemical equilibrium, the point at which the rates of the forward reaction and the reverse reaction are equal. We use the equilibrium constant, Kc, for a reaction to demonstrate whether or not the reaction favors products (the forward reaction is dominant) or reactants (the reverse reaction is dominant). High values of Kc mean that the reaction is product-favored, while low values of Kc mean that the reaction is reactant-favored.
EBooks Porter Stansberry Gold Investors Manual are currently available in various formats such as PDF, DOC and ePUB which you can directly download. In the Stansberry Research Gold Investor's Manual, you'll find several unique strategies to profit off this trend. And protect your wealth and your family in the. Investors manual for gold. The S&A Gold Investor's Manual on Amazon.com. *FREE* shipping on qualifying offers. You will find strategies to profit from investing in gold. And to protect your.
For acid and base dissociation, the same concepts apply, except that we use Ka or Kb instead of Kc. High values of Ka mean that the acid dissociates well and that it is a strong acid.
Low values of Ka mean that the acid does not dissociate well and that it is a weak acid. The same logic applies to bases. There is a relationship between the concentration of products and reactants and the dissociation constant (Ka or Kb). For acids, this relationship is shown by the expression: Ka = H3O+A- / HA. The products (conjugate acid H3O+ and conjugate base A-) of the dissociation are on top, while the parent acid HA is on the bottom. Notice that water isn't present in this expression. We get to ignore water because it is a liquid, and we have no means of expressing its concentration.
For bases, this relationship is shown by the equation Kb = BH+OH- / B. The products (conjugate acid and conjugate base) are on top, while the parent base is on the bottom. Once again, water is not present. Both the Ka and Kb expressions for dissociation can be used to determine an unknown, whether it's Ka or Kb itself, the concentration of a substance, or even the pH. Ka and Kb in Action Let's go into our cartoon lab and do some science with acids!
We need a weak acid for a chemical reaction. We have an acetic acid (HC2H3O2) solution that is 0.9 M. Its hydronium ion concentration is 4.
Animal Farm Study Guide Answers
10^-3 M. What is the Ka for acetic acid? Is this a strong or a weak acid? To solve this problem, we will need a few things: the equation for acid dissociation, the Ka expression, and our algebra skills.
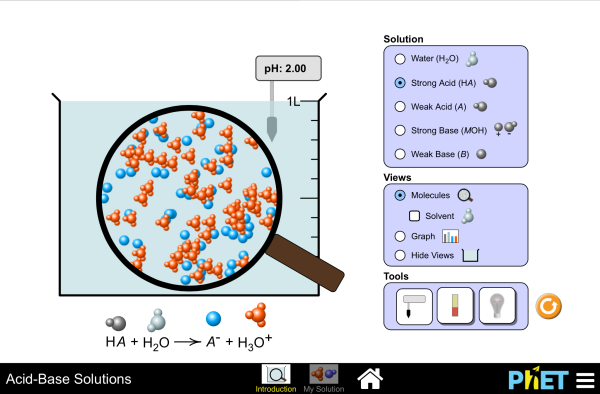
The equation is for the acid dissociation is HC2H3O2 + H2O H3O+ + C2H3O2. The Ka expression is Ka = H3O+C2H3O2- / HC2H3O2. The problem provided us with a few bits of information: that the acetic acid concentration is 0.9 M, and its hydronium ion concentration is 4. 10^-3 M. Since the equation is in equilibrium, the H3O+ concentration is equal to the C2H3O2- concentration. We plug the information we do know into the Ka expression and solve for Ka.
Ka = (4.0. 10^-3 M) (4.0. 10^-3 M) / 0.90 M Ka = 1.8. 10^-5 This Ka value is very small, so this is a weak acid. In another laboratory scenario, our chemical needs have changed. We absolutely need to know the concentration of the conjugate acid for a super concentrated 15 M solution of NH3.
We know that the Kb of NH3 is 1.8. 10^-5. Like with the previous problem, let's start by writing out the dissociation equation and Kb expression for the base. The equation is NH3 + H2O NH4+ + OH. NH4+ is our conjugate acid. Our Kb expression is Kb = NH4+OH- / NH3. We know that Kb = 1.8.
10^-5 and NH3 is 15 M. We can make the assumption that NH4+ = OH- and let these both equal x. The equation then becomes Kb = ( x)( x) / NH3. We plug in our information into the Kb expression: 1.8.
10^-5 = x^2 / 15 M. Solving for x, x = 1.6.
10^-2. Since we allowed x to equal NH4+, then the concentration of NH4+ = 1.6. 10^-2 M.
Finding pH Given Ka Here we are in the lab again, and our boss is asking us to determine the pH of a weak acid solution, but our pH probe is broken! The acid is HF, the concentration is 0.010 M, and the Ka value for HF is 6.8. 10^-4.
He has served as Scholar-in-Residence at The Kilns, Lewis's home in Oxford, and was a contributor to The Screwtape Letters: Annotated Edition and a member of the Advisory Board for The C. 

This assignment sounds intimidating at first, but we must remember that pH is really just a measurement of the hydronium ion concentration. We can find pH by taking the negative log of the hydronium ion concentration, using the expression pH = -log H3O+. Like in the previous practice problem, we can use what we know (Ka value and concentration of parent acid) to figure out the concentration of the conjugate acid (H3O+). Let's start by writing out the dissociation equation and Ka expression for the acid. HF + H2O H3O+ + F- The Ka expression is Ka = H3O+F- / HF. The concentration of H3O+ and F- are the same, so I replace them with x.
I put 6.8. 10^-4 for Ka, and 0.010 M for HF, then I solve for x. 6.8. 10^-4 = x^2 / 0.010 M x = 0.0026, so our hydronium ion concentration equals 0.0026 M. To find pH, I take the negative log of that. PH = -log 0.0026 pH = 2.6 Turns out we didn't need a pH probe after all. Lesson Summary Strong acids and bases dissociate well (approximately 100%) in aqueous (or water-based) solutions.
Weak acids and bases do not dissociate well (much, much less than 100%) in aqueous solutions. General acid dissociation in water is represented by the equation HA + H2O - H3O+ + A. General base dissociation in water is represented by the equation B + H2O - BH+ + OH. Ka and Kb values measure how well an acid or base dissociates. Higher values of Ka or Kb mean higher strength.
General Ka expressions take the form Ka = H3O+A- / HA. General Kb expressions take the form Kb = BH+OH- / B. When using Ka or Kb expressions to solve for an unknown, make sure to write out the dissociation equation, or the dissociation expression, first. Use the dissociation expression to solve for the unknown by filling in the expression with known information. Learning Outcomes Following this lesson, you should be able to:. Define strong and weak acids and bases. Explain what Ka and Kb values measure.
Identify the general Ka and Kb expressions. Recall how to use Ka and Kb expressions to solve for an unknown.
